Atsushi Fukunaga
Am J Clin Dermatol . 2022 Sep 15;1-14. doi: 10.1007/s40257-022-00728-6. Online ahead of print.
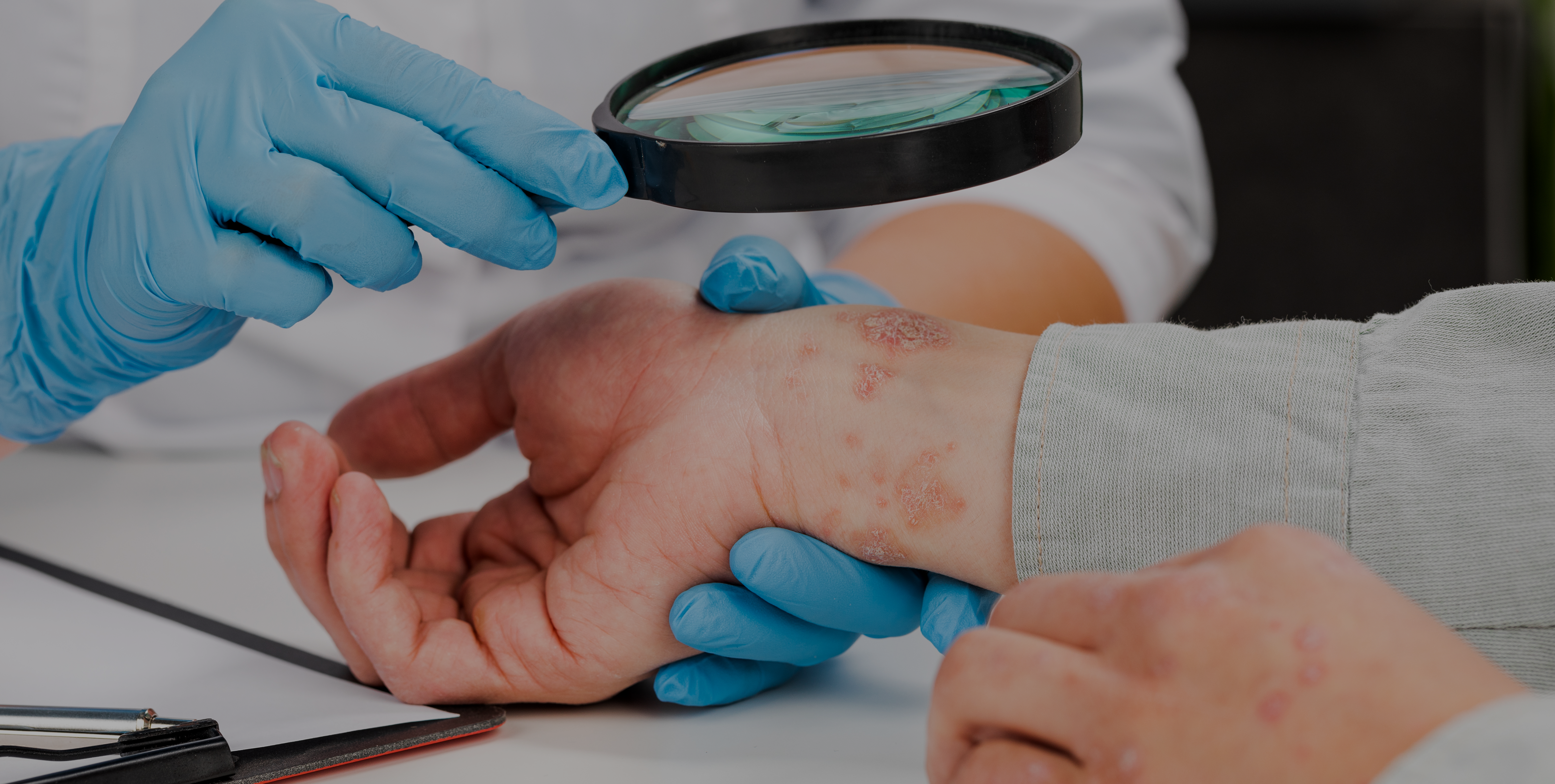
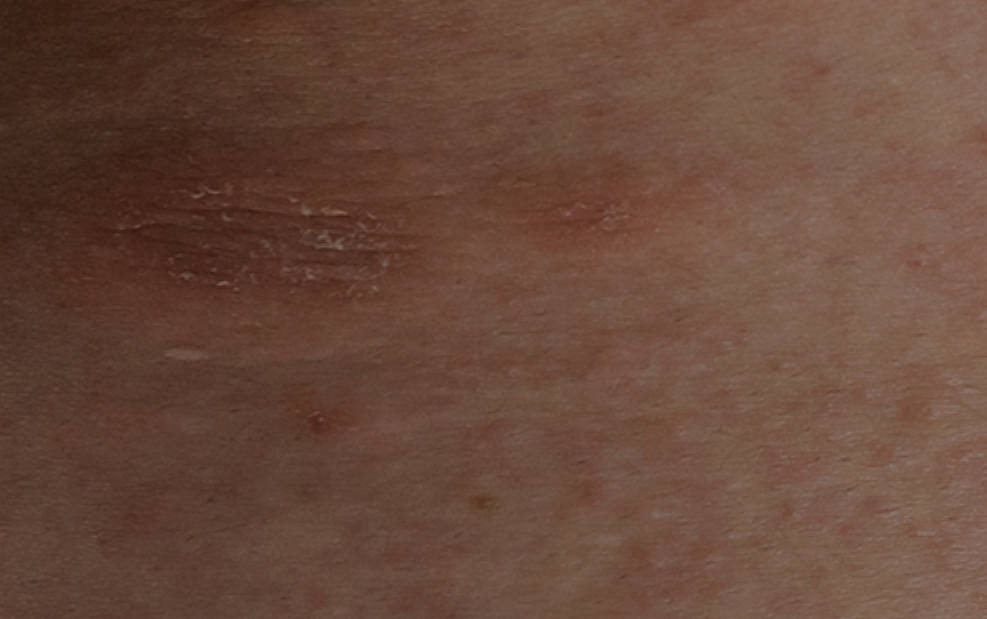
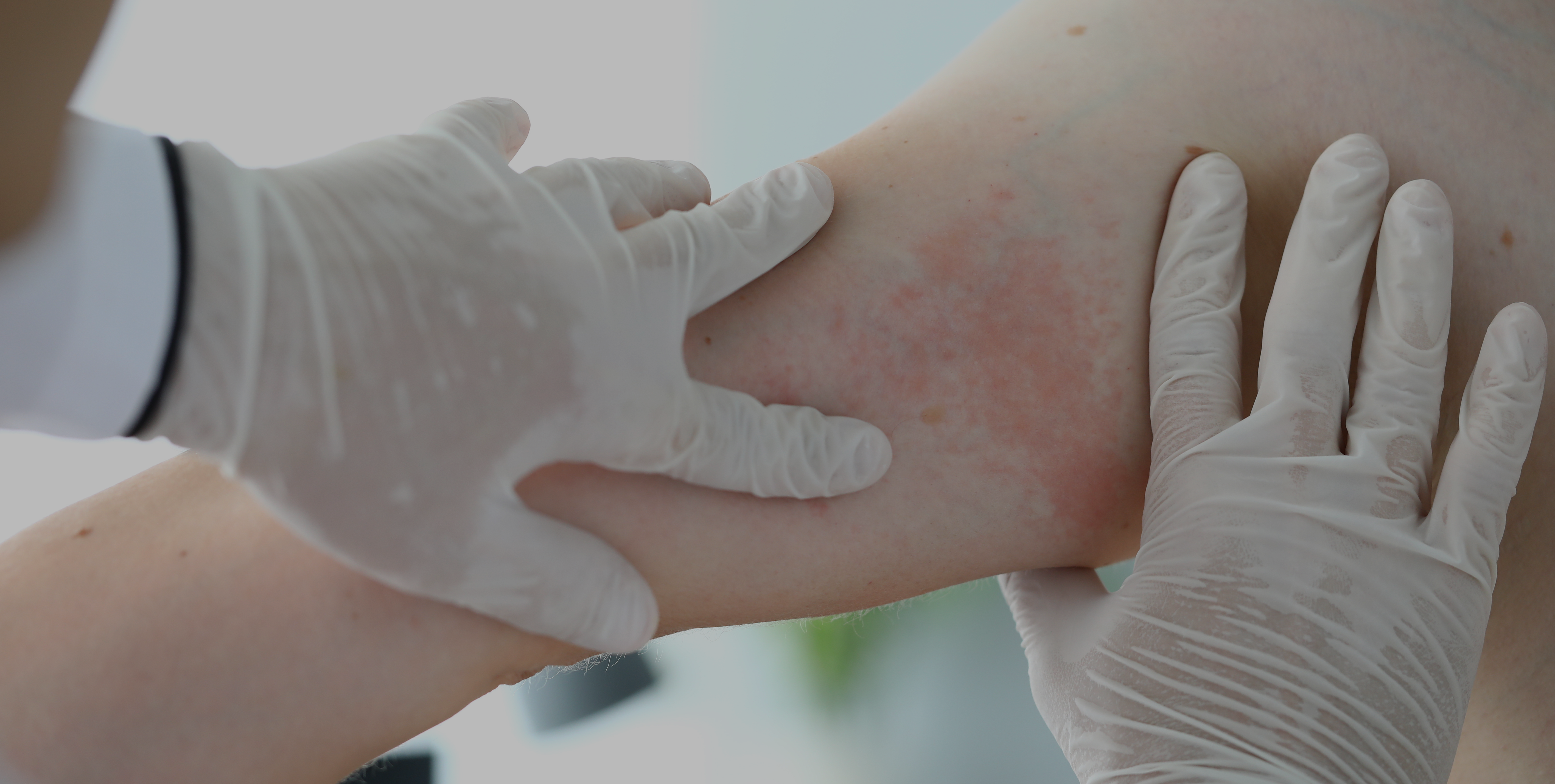
Cholinergic urticaria is a subtype of chronic inducible urticaria with painful wheals with surrounding erythema which occur after sweating. The objective of this review was to analyze different subtypes of cholinergic urticaria and its correlation to anhidrosis.
Recent publications were reviewed to update evidence on epidemiology, clinical features, diagnostic approaches, physiopathology subtype classification and therapeutic approaches of cholinergic urticaria.
When examining patients who present with symptoms similar to cholinergic urticaria, it is important to run a differential diagnosis to screen for a sweating dysfunction.
There are different mechanisms that contribute to the development of cholinergic urticaria, namely histamine, sweat allergy, cholinergic-related substances, poral occlusion and hypohidrosis/anhidrosis. These mechanisms are important to focus on the subtypes of cholinergic urticaria so that it can be managed and treated adequately, given the fact that this condition does not present in a homogeneous way.